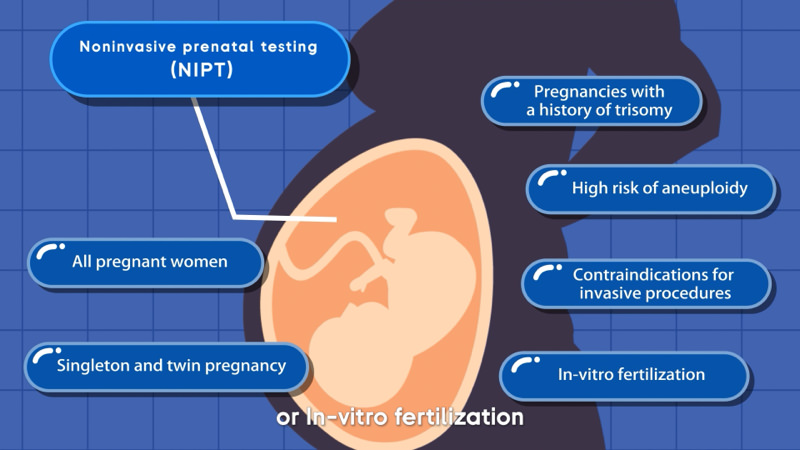
Overview of NIFTY®
According to American College of Obstetricians and Gynecologists (ACOG), cell-free DNA screening should be offered to all patients all pregnant patients regardless of maternal age or risk of chromosomal abnormality. Cell-free DNA screening is the most sensitive and specific screening test for common fetal aneuploidies1.

Conditions screen for :
- Trisomies (T21, T18, T13)
- Sex chromosomal aneuploidies
- Fetal sex

Conditions screen for :
- Trisomies (T21, T18, T13)
- Rare autosomal aneuplodies (T9, T16, T22)
- Sex chromosomal aneuploidies
- 84 types of Microdeletions/duplications syndromes
- Other aneuploidies
- Incidental Findings
- Fetal sex
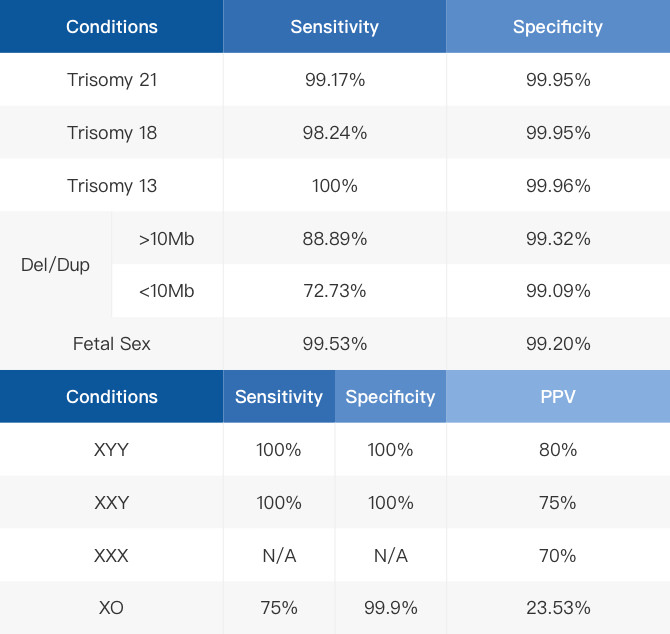
*The data in the table are based on past literature and Internal data[1],[2],[3],[4], and only reflect past testing conditions, and do not represent the actual situation or promised value of the tested samples.
BGI, a mature and
successful pioneer in NIPT
Largest test volume
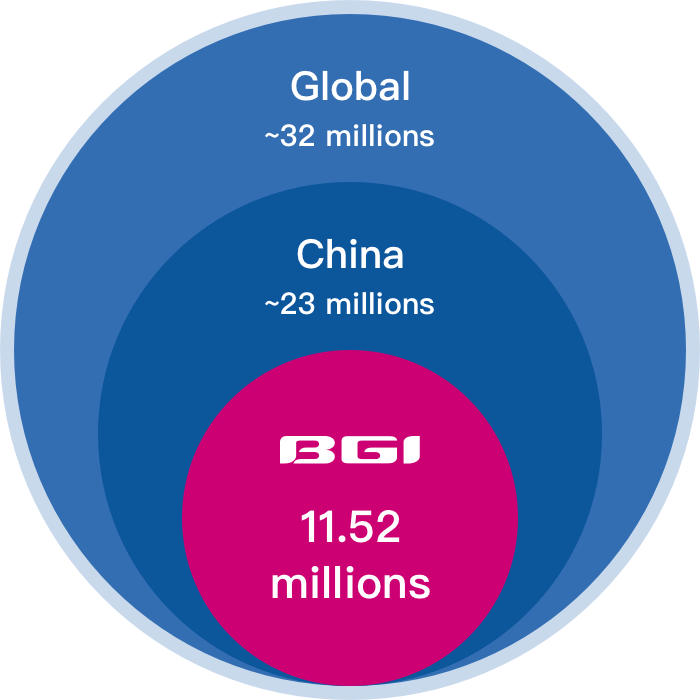
Excellent service verified by global market
• Validated in the largest prospective multicenter cohort NIPT study with 146,900+ pregnancies enrolled
• Trusted by 11,520,000+ family globally
• Serve in 100+ countries and regions
• 10+ years of professional experience in NIPT
• Performance evaluated in 70+ publications
• 10,000+ employees worldwide provide the most timely service
*Data updated in August 2022
NIFTY® advantages
-

Safe
Non-invasive with no risk of miscarriage
-

Simple
Test from a small >5ml maternal blood sample as early as week 10 of pregnancy
-

Accurate
Proven >99% sensitivity for T21, 18 & 13, based on a study of nearly 147,000 pregnancies
-

Trusted
Over 9,000,000 NIFTY® tests carried out to date by clinicians in more than 80 countries
-

Comprehensive
Screening options for 96 different genetic conditions

NIFTY® workflow
-
1

Conduct pre-test genetic counseling and ensure patient provides informed consent for test
-
2

Discuss and fill in the NIFTY® Consent Form/Test Request Form with the patient
-
3

Conduct blood draw
-
4

Send scanned copies of Consent Form/Test Request Form and information sheet to BGI
-
5

Arrange collection of the blood sample with a courier
-
6

Send Consent Form/Test Request Form with blood sample to BGI laboratory
-
7

Receive results back in 10 working days
-
8

Conduct pre- and post-test genetic counseling and provide drug guidance advice as required
Video
References:
1. Zhang H, Gao Y, Jiang F, Fu M, Yuan Y, Guo Y, Zhu Z, Lin M, Liu Q, Tian Z, Zhang H, Chen F, Lau TK, Zhao L, Yi X, Yin Y, Wang W. Non-invasive prenatal testing for trisomies 21, 18 and 13: clinical experience from 146,958 pregnancies. Ultrasound Obstet Gynecol. 2015 May;45(5):530-8. doi: 10.1002/uog.14792. Epub 2015 Apr 8. Erratum in: Ultrasound Obstet Gynecol. 2015 Jul;46(1):130. PMID: 25598039.
2.Liu H, Gao Y, Hu Z, Lin L, Yin X, Wang J, Chen D, Chen F, Jiang H, Ren J, Wang W. Performance Evaluation of NIPT in Detection of Chromosomal Copy Number Variants Using Low-Coverage Whole-Genome Sequencing of Plasma DNA. PLoS One. 2016 Jul 14;11(7):e0159233. doi: 10.1371/journal.pone.0159233. PMID: 27415003; PMCID: PMC4945049.
3. Song JP, Jiang YF, Gao TX, Yao YY, Liu LJ, Xu RH, Yi MQ, Yu CJ, Wang WP, Li H. Performance of non-invasive prenatal screening for sex chromosome aneuploidies and parental decision-making. Chin Med J (Engl). 2020 Jul 5;133(13):1617-1619. doi: 10.1097/CM9.0000000000000868. PMID: 32501830; PMCID: PMC7386330.
4.Jiang F, Ren J, Chen F, Zhou Y, Xie J, Dan S, Su Y, Xie J, Yin B, Su W, Zhang H, Wang W, Chai X, Lin L, Guo H, Li Q, Li P, Yuan Y, Pan X, Li Y, Liu L, Chen H, Xuan Z, Chen S, Zhang C, Zhang H, Tian Z, Zhang Z, Jiang H, Zhao L, Zheng W, Li S, Li Y, Wang J, Wang J, Zhang X. Noninvasive Fetal Trisomy (NIFTY) test: an advanced noninvasive prenatal diagnosis methodology for fetal autosomal and sex chromosomal aneuploidies. BMC Med Genomics. 2012 Dec 1;5:57. doi: 10.1186/1755-8794-5-57. PMID: 23198897; PMCID: PMC3544640.